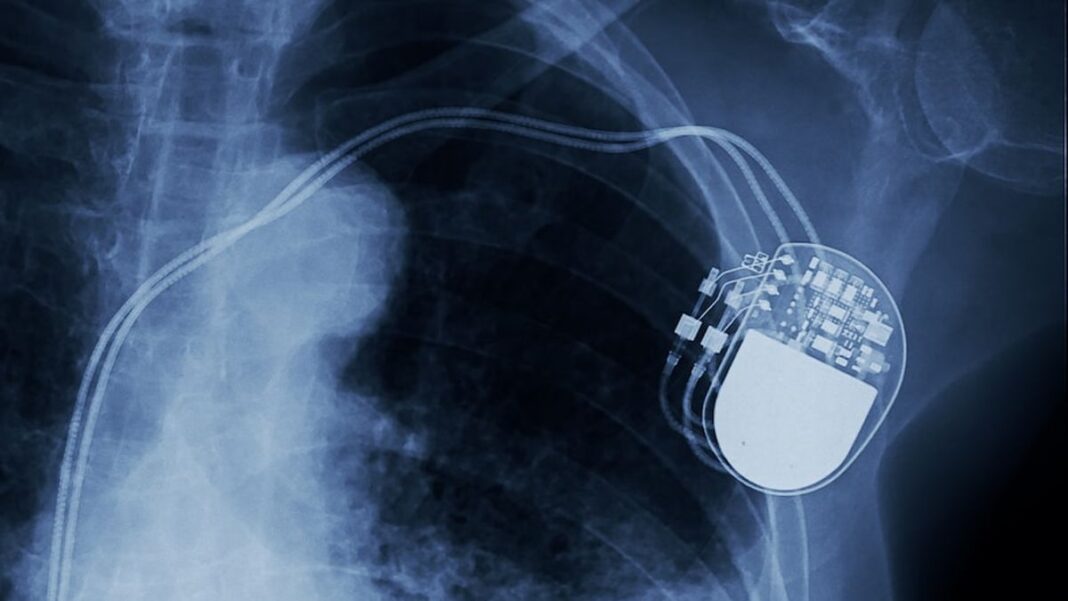
For decades, doctors have embedded pacemakers, cochlear implants and cardiac defibrillators into their patients’ bodies. More recently, consumers have started tracking their own heart rates and number of steps taken with watches, bracelets, cellphones and other wearable devices.
Researchers and doctors are now dreaming up more ways to merge those technologies, to move consumer-driven monitors inside bodies.
“We started to see this line blur with Fitbit,” said Amal Graafstra, who produces magnet implants and other technology through a company called Dangerous Things, which he founded in 2013. “When you start looking into medical applications, there’s going to be a convergence, and I think that’s going to be inevitable. ”In some ways, this is already happening: The medical field has fast taken to microchip implants, ultra-slim and flexible digital tags that can, for example, control a prosthetic limb. Such technology can also monitor the movements of Parkinson’s patients using accelerometers to detect tremors, which offers neurologists a detailed assessment of a patient’s physical behavior before they visit for their next appointment, aiding in the selection of appropriate doses of medicine. Researchers at the University of Illinois developed electronic sensors that can be embedded into temporary skin tattoos to track and stimulate brain waves to prevent seizures.
This technology offers the potential for screening before an appointment without having to send a patient to a lab for testing. It could also allow patients to always carry their medical records.
“The analytical laboratories will continue to do their job. But with the lab-on-chip . . . general practitioners and paramedics will have a tool at their disposal to carry out tests on the patient before going to the laboratory,” said Giuseppe Barillaro, an associate professor of electronics at the University of Pisa and the co-author of a study on bioabsorbable materials, which aid in healing and fully dissolve once the implant is no longer needed. He said that the technology is still maturing and the commercial availability for broad use is not available for all applications.
As health-care costs continue to rise, hospital stays have often been shortened and researchers are exploring the potential for ambulatory monitoring.
An example of this: A continuous glucose monitoring implant for adults with diabetes that pairs with a mobile app. It was first approved by the Food and Drug Administration in 2018. The system was created by Eversense and is operational for 90 days. The company hopes to create a sensor with 365 days of wearable time.
For now, users still need to prick their fingers for blood samples twice daily to calibrate the device, which also vibrates to warn patients of an impending or actual hypoglycemia or hyperglycemia event.
The implant, a small and thin white circular device, is externally located on the patient’s shoulder where a quick tap of a cellphone can give instant readings. The FDA evaluated clinical study data from 125 diabetics who used the device and found less than 1 percent of users experienced a “serious adverse event.”
Mirasol Panlilio, a spokesperson for the company, said that users have expressed a desire to make the device removable, if only to allow them a “naked shower” with nothing attached to their body, something the company is pursuing in future versions of the implant.
The tech has been slow to develop as fewer volunteers are willing to sign up to clinical studies, unlike vaccine pathways that draw hundreds of thousands willing to test preliminary doses, said Hubert H. Lim, a professor who researches implantable medical tech at the University of Minnesota’s Department of Biomedical Engineering.
Until now, Lim said hobbyists seeking to “hack” their bodies, like installing a magnet or the key to a Tesla in a hand, are the most willing to take the risks and try implanted devices. Implantable tech could cause trouble in situations where an individual needs an MRI scan or airport security scanners, he said.
By Kenneth R. Rosen
See Read Full Article on WashingtonPost.com