What Did Pfizer Know, and When Did They Know It? Neurological Harms Concealed.
This report assists in answering, “What did Pfizer know, and when did they know it?” concerning its COVID-19 vaccine. The report focuses on neurological complaints post-injection with the Pfizer COVID-19 vaccine, as well as on several other, non-neurological reported symptoms.
The information presented comes from the Centers for Disease Control and Prevention (CDC) Wonder website (CDC.Wonder.gov) through which anyone can access CDC’s VAERS system. VAERS is a reporting system for vaccine manufacturers, health care providers, and the general public to notify the CDC of issues, injuries, symptoms, any problem with a vaccine.
The Vaccine Adverse Event Reporting System (VAERS) provides answers to what Pfizer knew about vaccine injuries resulting from its COVID-19 vaccine and when they knew it. The purpose of VAERS is to alert Pfizer, the CDC, and the Food and Drug Administration (FDA) to safety signals requiring investigation.
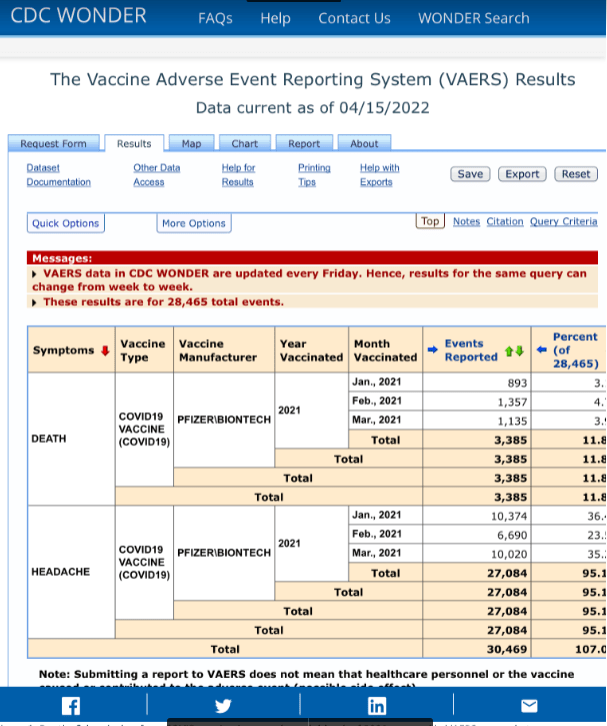
Below are seven screenshots of six VAERS reports obtained directly from the VAERS system.
1) The first screenshot shows reports of deaths and headaches reported by those vaccinated in January, February, and March of 2021. The mass vaccination of Americans had just started in that time frame. VAERS reports from the first three months gave Pfizer, the CDC and the FDA critical safety signal information to act upon, though they chose not to address the clear safety signals.
This screenshot shows 3,385 deaths reported in three months, as well as 27,084 headaches which will be elaborated upon in another screenshot. [https://wonder.cdc.gov/controller/datarequest/D8;jsessionid=6227282DDE2B9107FA07D6EF49E0]